PDF) Corrective and Preventive Action (CAPA) and Complaint Handling in Medical Device. - By Compliance Global Inc | Compliance Global Inc - Academia.edu

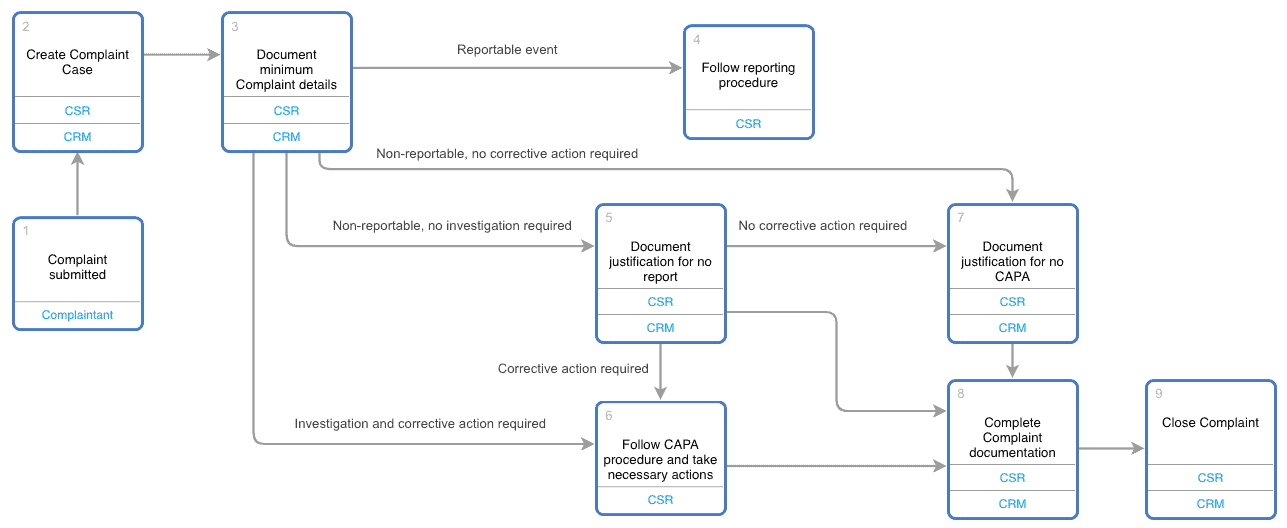
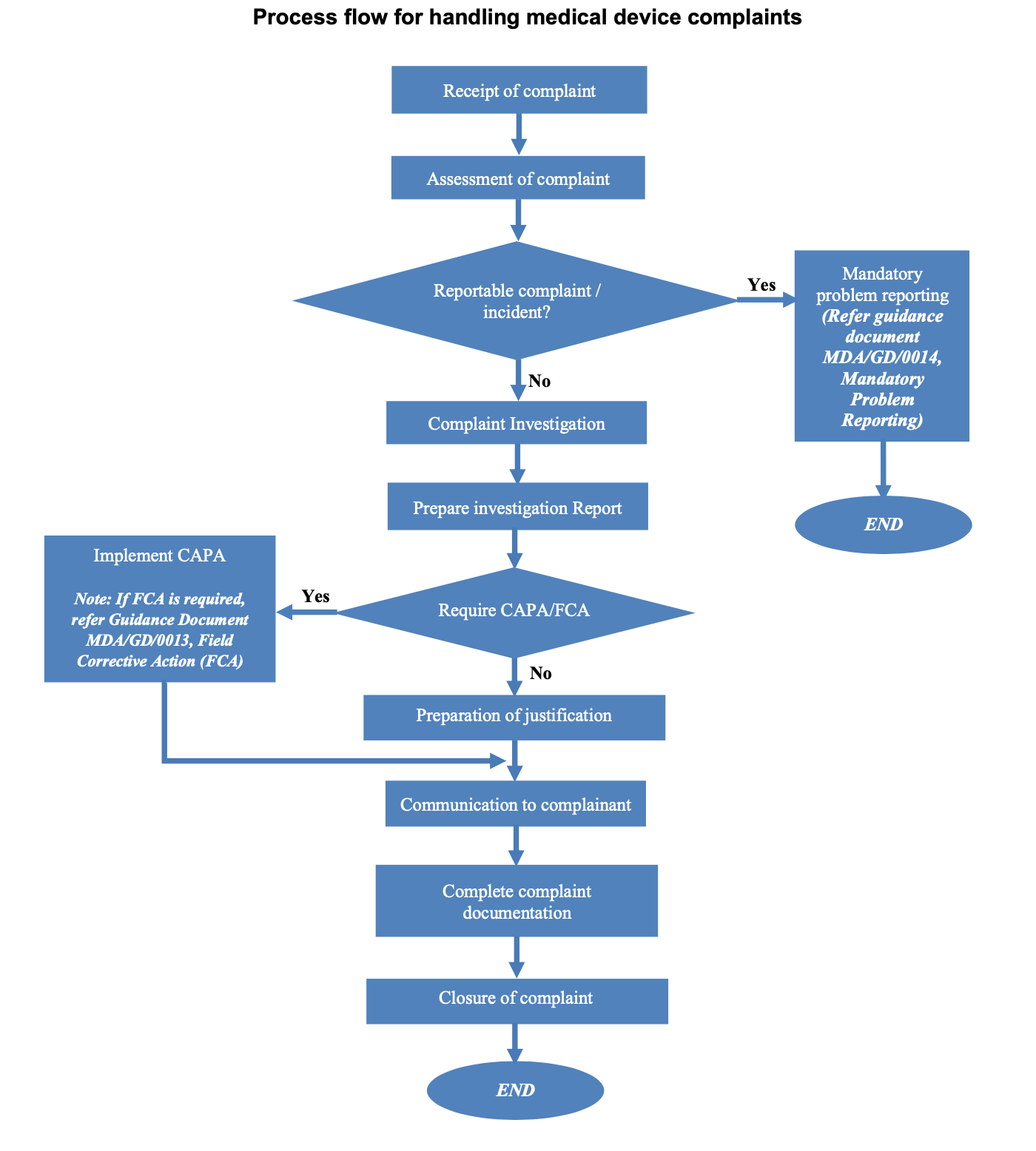
Complaint Handling Requirements - Interrelationship with CAPA, Change Control, Adverse Event Reporting and Recalls, Life Cycle Process Activities - Webinar Compliance


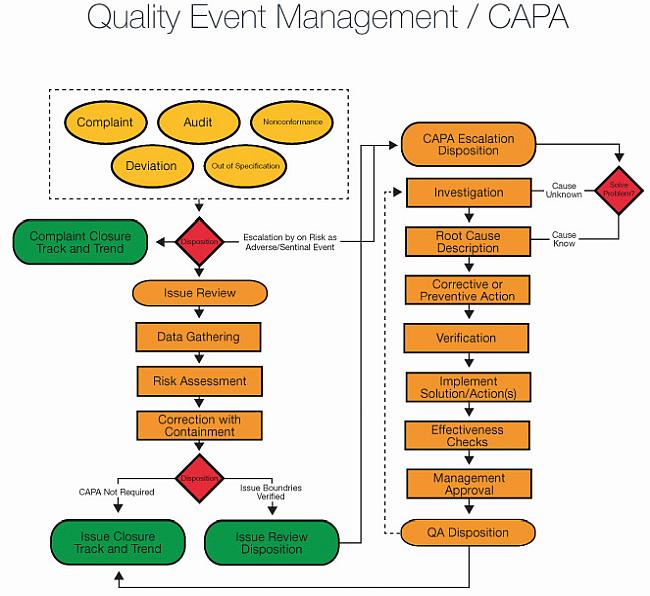
![CAPA Process: Step-by-Step Instructions [+ Free Infographic] CAPA Process: Step-by-Step Instructions [+ Free Infographic]](https://blog.greenlight.guru/hubfs/capa2.png)